Document Type
Article
Publication Date
May 2007
Abstract
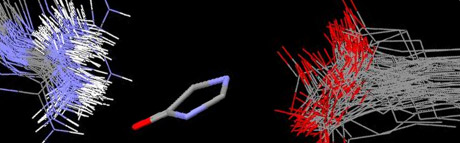
In the ground state of the highly conjugated green fluorescent protein (GFP), the chromophore should be planar. However, numerous crystal structures of GFP and GFP-like proteins have been reported with slightly twisted chromophores. We have previously shown that the protein cavity surrounding the chromophore in wild-type GFP is not complementary with a planar chromophore. This study shows that the crystal structure of wild-type GFP is not an anomaly: most of the GFP and GFP-like proteins in the protein databank have a protein matrix that is not complementary with a planar chromophore. When the π-conjugation across the ethylenic bridge of the chromophore is removed the protein matrix will significantly twist the freely rotating chromophore from the relatively planar structures found in the crystal structures. The possible consequences of this non-planar deformation on the photophysics of GFP are discussed. In addition to GFP there are GFP-like proteins that can be reversibly photoswitched between a fluorescent and a nonfluorescent state. Conformational searching and molecular dynamics are used to examine the various kindling proteins with a cleaved chromophore. MD simulations supported that the chromophore does indeed start to move away from its initial trans configuration, and actually completes a full trans/cis isomerization. The nearby His197 residue does not provide a large barrier to the chromophore rotation when it is cleaved from the protein backbone. This implies that the rest of the protein backbone did not provide an obstacle to the isomerization. Fusion of the chromophore back to the protein backbone was also done and conformational analyses showed that the proteins with fused and cleaved chromophores did now show a great deal of change the φ and τ dihedral angles most cases.
Recommended Citation
Maddalo, Scott L., "The Effect of the Protein Matrix on Fluorescence and an Analysis of the Effect of Fusing Residues 62 and 63 in Kindling Proteins" (2007). Chemistry Honors Papers. 2.
https://digitalcommons.conncoll.edu/chemhp/2
The views expressed in this paper are solely those of the author.